|
Back to Blog
Western blot test lyme5/17/2023  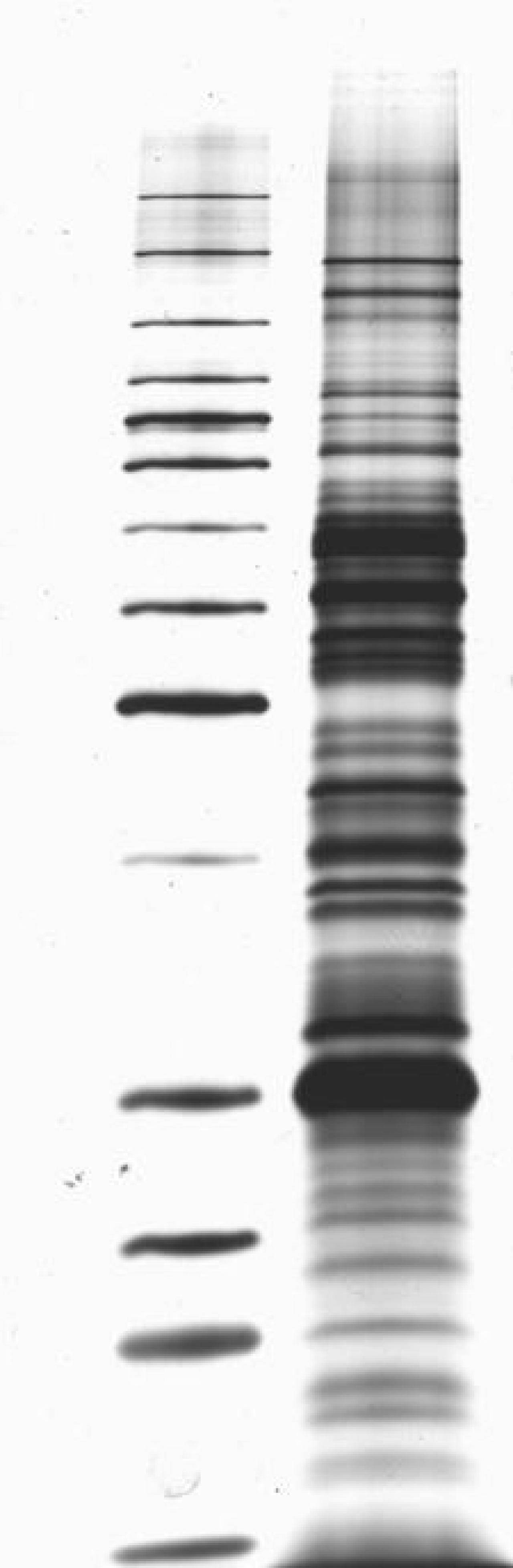
This kind of approach, which has been approved by the FDA for one company (though I’m sure others will seek approval as well) ultimately allows for a faster and likely less expensive diagnostic approach to Lyme disease testing. They are a difficult technology and a bit more variable. Further, laboratories do not like doing immunoblots. 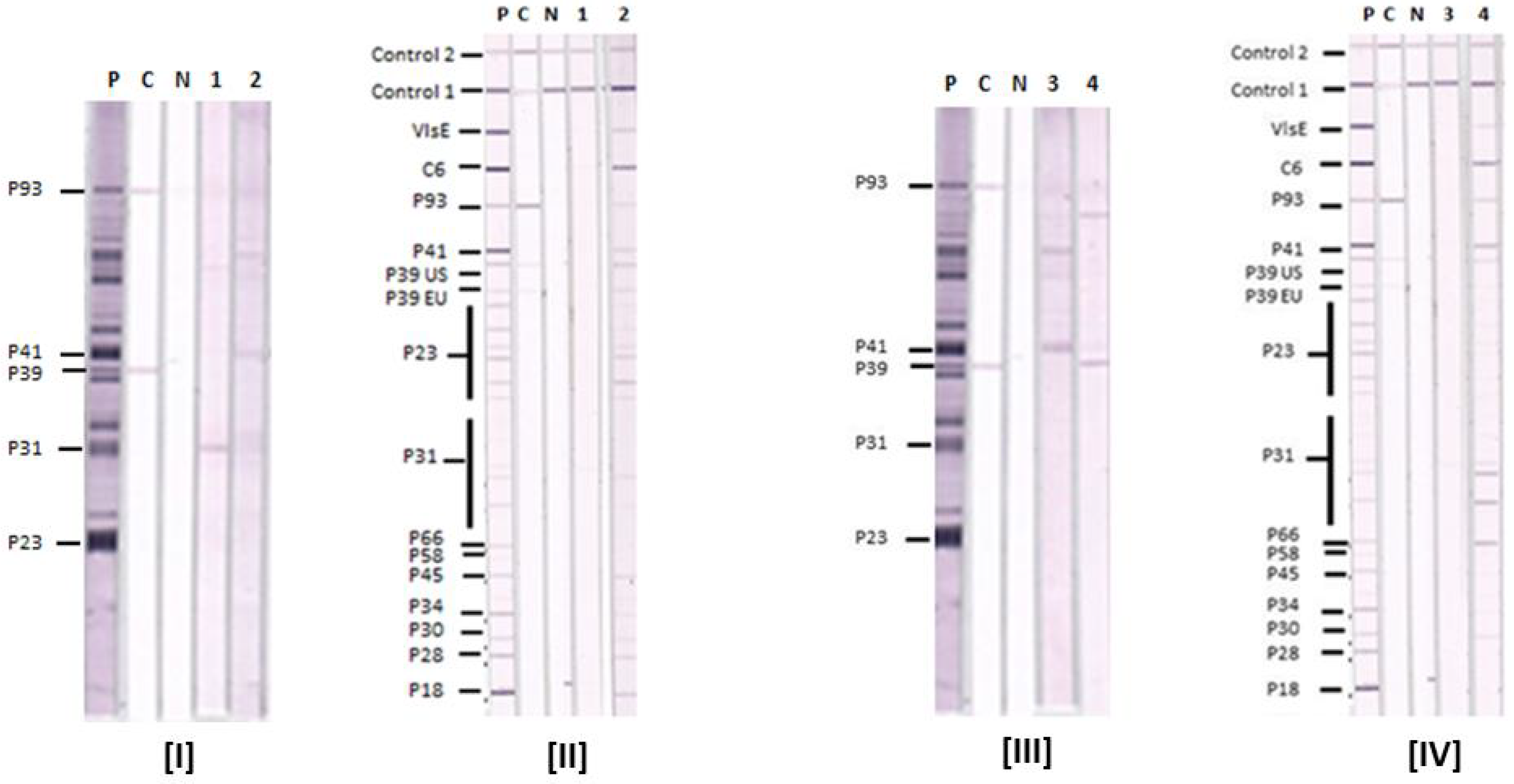 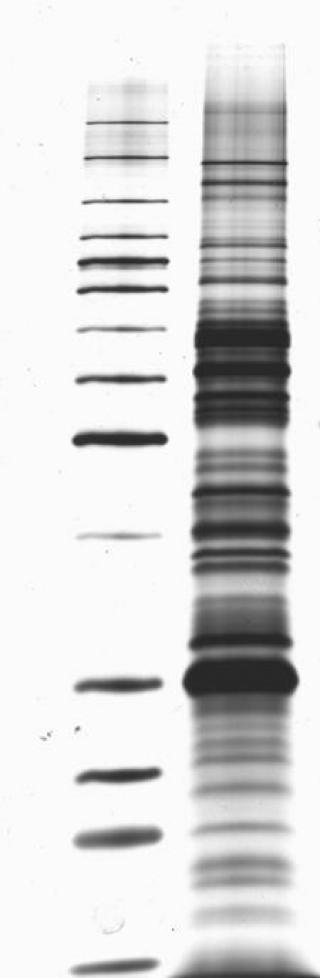
The reporting of positive or negative tests can usually be accomplished within a day or two, at most. Some of these modified two-tier tests use antigens that allow for earlier diagnosis after Lyme infection than standard tests. You get results quickly the same day or the next day. These are platforms that most labs can run in-house. In the newer, two-tier test, two EIAs are done either at the same time or sequentially. That methodology in most clinical settings means you don't get a result back for 3 to 7 days after obtaining a blood test because immunoblots usually take time to be batched and run. If that is positive, you then do immunoblots - a bit of an old-fashioned technology - to look for specific antibodies against antigens. This includes a first-tier screen with an EIA. The current serologic testing for Lyme disease is a so-called two-tier test. Neither test is meant to be a screening test, and they should not be ordered for patients with a low prior probability of Lyme disease, such as patients with only nonspecific symptoms. Of course, the predictive value of a positive result (for either the old or the new tests) are highly dependent on the prior probability that the patient has Lyme disease. Although preliminary data on the sensitivity and the specificity of the new tests are promising, the true values will not be known until they have been used widely. The new tests should be less expensive than testing with an ELISA and western blots, and the results should be available sooner. The newly approved diagnostic test system is a different two-tiered testing algorithm in which both the first-tier and second-tier tests are ELISAs that measure different antigens of B. 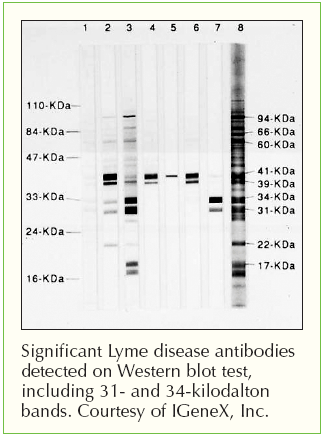
However, performing immunoblots is both time-consuming and expensive, and it is difficult to eliminate at least some degree of subjectivity in interpreting the blots. This testing algorithm has proved to be very good (except for early infection, because erythema migrans typically develops before antibodies are detectable). The result is positive only if both the ELISA and the western immunoblot results are positive. If that result is either positive or equivocal, then a second-tier test, a western immunoblot, is performed. The first is a quantitative test, typically an enzyme-linked immunoassay (ELISA). Recommended testing for antibodies to Borrelia burgdorferi entails a two-tier algorithm. Under the new recommendation, when cleared by the FDA for Lyme disease testing, “serologic assays that utilize EIA rather than western immunoblot assay in a two-test format are acceptable alternatives for the laboratory diagnosis of Lyme disease.”Īccording to the update, “ clinicians and laboratories should consider serologic tests cleared by FDA as CDC-recommended procedures for Lyme disease serodiagnosis” - a standard based on the criteria established at the 1994 Second National Conference on Serologic Diagnosis of Lyme Disease. The CDC previously recommended a two-tier testing algorithm that included a sensitive first-tier test - either an EIA or immunofluorescence assay - followed by a supplemental immunoblot assay if the first test resulted in a positive or equivocal result. The update, published in MMWR, followed the FDA’s clearance last month of four previously cleared Lyme disease tests manufactured by ZEUS Scientific with new indications for use in the modified two-tier testing process. The CDC issued an updated recommendation for the serologic diagnosis of Lyme disease, endorsing the use of a two-tier testing algorithm that uses a second enzyme immunoassay, or EIA, in place of a western immunoblot assay. If you continue to have this issue please contact to Healio 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
